About us
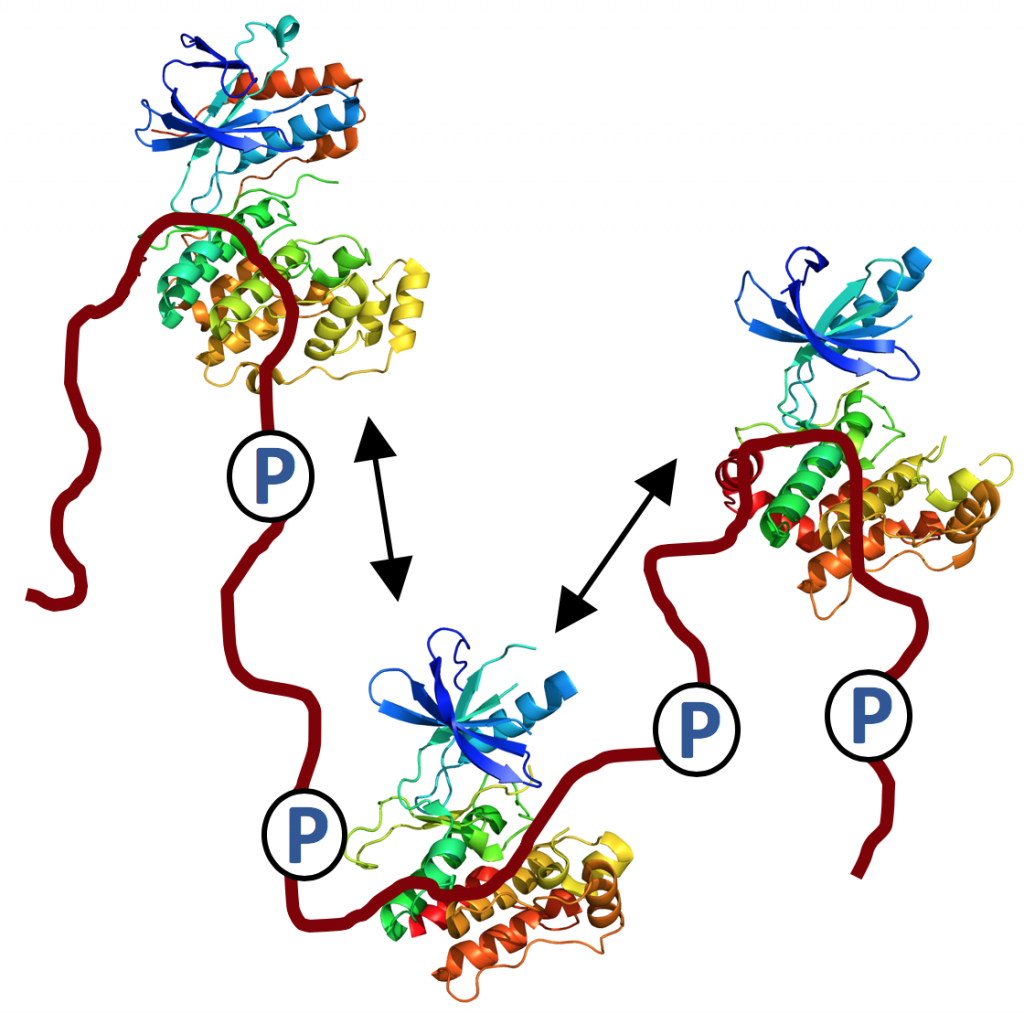
Our research team focuses on elucidating the role of intrinsically disordered proteins (IDPs) in biology. In particular, we are interested in the mitogen-activated protein kinase (MAPK) cell signaling pathways and how intrinsically disordered scaffold proteins regulate signaling specificity and MAPK activity. We use NMR spectroscopy combined with X-ray crystallography and biophysics to study these dynamic assemblies at atomic resolution.
Latest news
03/2024 Stefan Nebl joins the team as an Impulscience postdoctoral fellow
11/2023 Marion Chenal joins the team as an Impulscience Ph.D. student
10/2023 Thomas Winbolt joins the team as a Ph.D. student
06/2023 Maud Tengo joins the team as an Impulscience research technician
01/2023 Elise Delaforge joins the team as an Impulscience postdoctoral fellow
11/2022 Malene is laureate of the Impulscience program of the Bettencourt Schueller foundation. Thanks for the amazing support. Great research ahead.

See more news here
